What Exactly is “Cold Chain Leakage” ?
Cold chain leakage refers to any unintended deviation from the required temperature range at any point in the storage and distribution journey — from manufacturer to end consumer. It is not merely about a broken refrigerator. Leakage happens structurally, operationally, and architecturally.
The Five Root Causes of Cold Chain Leakage:
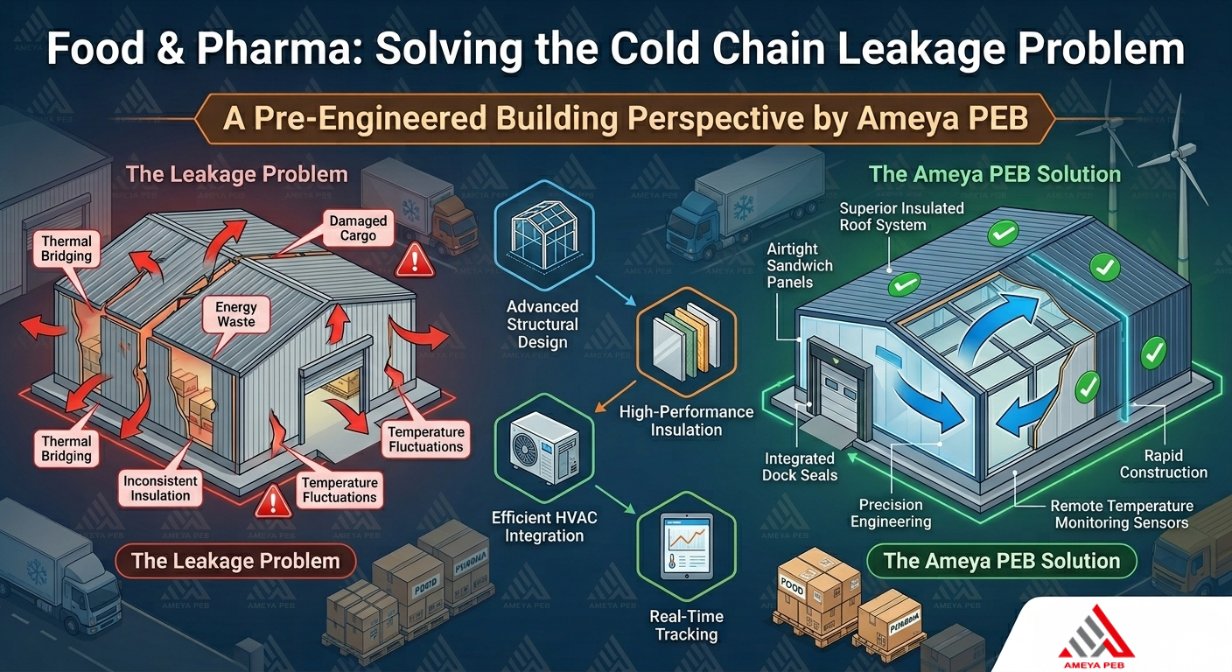
1. Thermal Bridging in Building Envelopes: Poor insulation design and gaps in the building structure allow heat to infiltrate cold rooms, forcing refrigeration systems to overwork and creating micro-zones of uneven temperature distribution.
2. Door & Loading Bay Vulnerabilities: Each loading dock interaction causes a surge of ambient warm air. Without precision-engineered vestibules and rapid-roll doors, temperature recovery time stretches — leaving products exposed.
3. Vapour & Moisture Infiltration: Inadequate vapour barriers in walls and floors lead to condensation, icing on floors, frost buildup on evaporator coils, and biological contamination risks for food and pharma products.
4. Inefficient Refrigeration Load: When structural insulation is poor, refrigeration systems carry a disproportionate load — increasing energy costs by 30–50% and reducing equipment lifespan significantly.
5. Regulatory Non-Compliance: Pharmaceutical cold stores must meet WHO GDP, FSSAI, and Schedule M requirements. Structurally deficient buildings routinely fail audits, leading to product recalls, license risks, and reputational damage.
| Critical Insight: Studies show that over 65% of cold chain temperature excursions originate not from refrigeration equipment failure — but from structural inadequacies in storage buildings themselves. The building IS the cold chain. |
Why Conventional Construction Fails the Cold Chain
Traditional brick-and-mortar or RCC-framed cold storage facilities were never engineered with precision thermal performance as a first principle. They are retrofitted with insulation as an afterthought — applied to walls not designed for it, joints not sealed properly, and roofs that develop thermal bridges over time.
The consequences are structural and cumulative. Every temperature excursion adds to regulatory risk, product spoilage, energy bills, and refrigeration maintenance costs. For pharmaceutical companies operating under WHO-GDP or Schedule M frameworks, a single non-conformance can halt an entire product batch worthcrores.
Conventional Construction vs. PEB Cold Chain: A Comparison
| Parameter | Conventional Construction | PEB Cold Chain Facility |
| Thermal Performance Design | ✗ Retrofit insulation | ✓ Engineered from foundation |
| Construction Time | ✗ 18–36 months | ✓ 4–8 months |
| Vapour Barrier Continuity | ✗ Inconsistent | ✓ Continuous, engineered |
| Structural Flexibility | ✗ Difficult & costly | ✓ Modular, expandable |
| Regulatory Audit-Readiness | ✗ Case by case | ✓ Designed to comply |
| Energy Efficiency | ✗ High leakage load | ✓ Optimized thermal envelope |
| Long-term Maintenance | ✗ High (moisture, cracking) | ✓ Low (sealed, durable) |
The PEB Advantage: Engineering Cold, Not Fighting It
Pre-Engineered Buildings (PEB) represent a fundamental shift in how cold storage infrastructure is conceived. Rather than building a generic structure and then attempting to make it cold-capable, PEB starts with the thermal requirement and engineers the entire structure — steel frame, insulated panels, roof, loading areas, and utilities — as an integrated system.
| Industry Perspective: A cold chain facility isn't a warehouse with a refrigerator. It's a precision instrument — and it must be built like one. |
Six Engineering Pillars of PEB Cold Storage
High-Performance Insulated Panels PUF or PIR sandwich panels with precisely controlled core density — achieving U-values as low as 0.18 W/m²K, dramatically reducing heat ingress. | Thermal Break Steel Connections Standard steel connections are notorious thermal bridges. PEB designs use thermal break elements at every structural joint to eliminate heat infiltration pathways. |
Continuous Vapour Barrier Systems Every panel joint, penetration, and corner is factory-detailed and site-executed to form an uninterrupted vapour barrier — protecting both structure and stored products. | Engineered Loading Bay Airlocks Dock levellers, dock shelters, rapid-roll doors, and thermal vestibules control air exchange during loading — the most thermally vulnerable operational moment. |
Cool Roof & Solar-Ready Design Specialised roof coatings with high solar reflectance indices (SRI) reduce rooftop heat gain by up to 30%, directly reducing compressor load and operating costs. | Compliance-Ready Layouts For pharma clients, PEB layouts incorporate GDP-mandated segregation zones, quarantine areas, sampling rooms, and controlled access — designed in, not added on. |
| Sector Deep-Dive: Food & Beverage Cold Chain |
India is one of the world's largest producers of fruits, vegetables, dairy, meat, and seafood. Yet post-harvest losses across these categories remain among the highest globally — a direct consequence of cold storage infrastructure that is either unavailable, inadequate, or unreliable.
Multi-Temperature Zones in a Single Structure
Modern food processing and distribution requires multiple temperature regimes within a single campus: deep freeze (-25°C to -18°C) for meat and ice cream; chilled storage (0°C to 5°C) for dairy and fresh produce; and controlled atmosphere rooms for fruits and vegetables. PEB structures are uniquely capable of delivering multi-temperature facilities within a shared footprint, with precisely insulated dividing walls preventing zone crossover.
HACCP & FSSAI Compliance by Design
Food safety frameworks require hygienic wall-floor junctions, washdown-capable surfaces, pest-proof construction, and air filtration systems. PEB panel systems with coated steel facings and sealed joints are designed to meet HACCP requirements as a baseline, not an upgrade.
| Result: Food cold stores built with PEB methodology report 20–35% lower energy consumption compared to conventional RCC structures of equivalent capacity — while achieving higher thermal consistency and faster audit clearance. |
| Sector Deep-Dive: Pharmaceutical Cold Chain |
The pharmaceutical cold chain operates under the most stringent regulatory scrutiny of any temperature-controlled environment. Whether for biologics, vaccines, active pharmaceutical ingredients (APIs), or finished dosage forms, the margin for temperature excursion is measured in fractions of a degree — and the consequences of failure are measured in patient safety.
GDP & Schedule M Requirements
WHO Good Distribution Practice (GDP) guidelines and India's Schedule M requirements mandate continuous temperature monitoring, validated mapping studies, defined SOPs for excursion management, and documented qualification of storage areas. All of these requirements place extraordinary demands on the physical structure — which must maintain conditions independently of procedural compliance.
The Qualification Advantage of PEB
PEB pharma cold stores are engineered with factory-traceable panel specifications, enabling IQ/OQ/PQ (Installation, Operational, and Performance Qualification) studies to proceed with documented baseline data. This reduces qualification timelines, supports faster regulatory submissions, and provides the audit trail that inspectors require.
Pharma Storage Categories & PEB Specifications
| Storage Category | Temp Range | PEB Panel Specification |
| Ambient (CRT) | 15°C – 25°C | 50mm PUF insulated wall & roof panels |
| Cold Room | 2°C – 8°C | 100mm PIR panels, thermal break connections |
| Freezer Room | -20°C ± 5°C | 150mm PIR, continuous vapour barrier, heated floor |
| Ultra-Low Temperature | -70°C to -40°C | 200mm+ PIR, cryogenic-grade seals, anti-condensation systems |
FEATURED PARTNER Ameya PEB: Engineered for Every Degree Ameya PEB is India's specialist in pre-engineered building solutions for temperature-sensitive industries. With deep expertise in cold chain infrastructure for food processing, pharmaceutical distribution, and bulk chemical storage, Ameya PEB delivers facilities that are not just built — they are precision-engineered to maintain product integrity from day one to decade ten. What sets Ameya PEB apart is an integrated design philosophy: thermal engineering, structural design, MEP coordination, and regulatory compliance are unified in every project — from the first drawing to the final qualification certificate. |
| ISO Certified Design & Manufacturing | GDP-Ready Pharma Layouts | Pan-India Project Execution |
Ameya PEB's Cold Chain Building Process
Ameya PEB follows a structured, client-aligned process that translates cold chain requirements into engineering specifications — and then into compliant, operational facilities.
Step 1: Thermal & Regulatory Needs Assessment
Every project begins with a detailed review of the client's product profiles, temperature requirements, throughput volumes, and applicable regulatory frameworks (FSSAI, WHO-GDP, Schedule M, NABH). This shapes all downstream engineering decisions.
Step 2: Integrated Thermal & Structural Design
Ameya's engineering team conducts heat load calculations, selects panel specifications, designs thermal break connections, and coordinates HVAC & refrigeration integration — all within a unified BIM-informed model.
Step 3: Factory Fabrication & Quality Control
Structural steel is fabricated in controlled factory conditions to precise tolerances. Panel systems are procured from certified manufacturers with traceable specifications — ensuring consistency that site-built alternatives cannot match.
Step 4: Precision Site Erection
Ameya PEB's supervised erection teams assemble the structure with a focus on airtightness — every panel joint is sealed, every penetration is thermally detailed, and every connection is thermal-break compliant.
Step 5: Commissioning, Mapping & Handover
Post-construction, Ameya PEB coordinates temperature mapping studies, supports equipment commissioning, and provides full as-built documentation packages to support client qualification programs.
The Economic Case: Beyond Capital Cost
Decision-makers evaluating cold storage investments often focus on upfront capital expenditure — and on this dimension alone, PEB already offers advantages through faster construction and reduced civil work. But the true economic case for PEB in cold chain applications is built over the asset's life:
Energy Cost Reduction Superior thermal envelope performance directly reduces refrigeration compressor run-time. PEB cold stores consistently achieve 20–40% lower energy consumption than equivalent conventional structures. | Lower Maintenance Costs Sealed, non-porous panel surfaces resist moisture, mould, and structural degradation. Maintenance requirements and associated facility downtime are significantly lower over a 20–30 year lifecycle. |
Reduced Product Loss For food clients, even a 5% reduction in spoilage rates translates into crores of recovered value annually. Precise temperature control directly drives product shelf life and marketable yield. | Audit & Compliance Savings Pharma clients avoid costly regulatory non-conformances, product recalls, and re-qualification cycles — all of which carry direct financial penalties and reputational risk far exceeding any premium in PEB construction cost. |
| Lifecycle Perspective: A PEB cold store designed to last 30 years with high thermal performance will typically recover its premium over conventional construction within 4–7 years through energy savings alone — before counting product loss reduction or regulatory avoidance. |
Future-Proofing India's Cold Chain
India's cold chain sector is at an inflection point. Government initiatives like the Pradhan Mantri Kisan Sampada Yojana and the PLI schemes for food processing are channelling significant capital into cold chain expansion. Simultaneously, the pharmaceutical sector's post-pandemic growth has created unprecedented demand for GDP-compliant cold storage — particularly for biologics, cell therapies, and temperature-sensitive vaccines.
In this environment, the facilities being built today will define cold chain performance for the next two to three decades. Choosing construction methods that embed thermal performance as a core engineering principle — rather than a retrofit — is not merely a technical decision. It is a strategic one.
Pre-Engineered Buildings, designed and delivered by specialists like Ameya PEB, represent the most reliable path to cold chain infrastructure that performs as specified, complies as required, and operates as economically as possible — for the entire life of the asset.